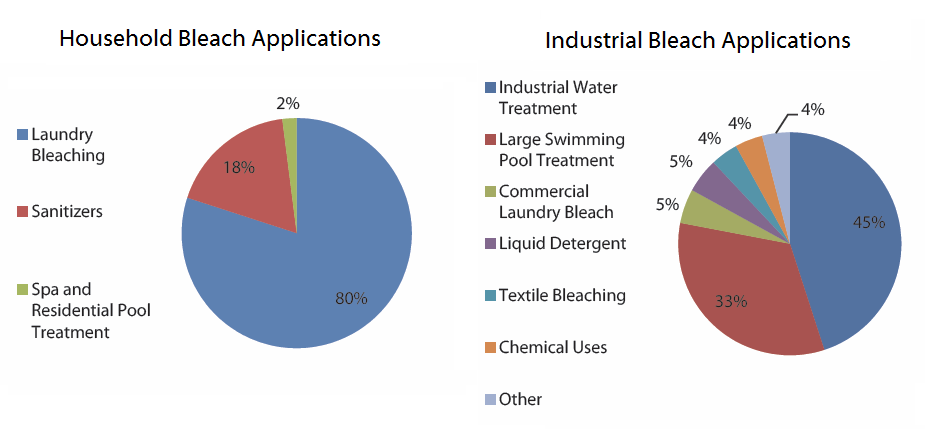
The usages of Sodium Hypochlorite (Bleach) is divided into two types of categories: Household Bleach and Industrial Bleach. ("Intratec Solutions LLC", 2005)
Although the usages of bleach is divided into two categories; Household Bleach and Industrial Bleach, the common and general purposes of using sodium hypochlorite is for purification and bleaching. (Schlager, Weisblatt, & Newton, 2006)
1. Disinfectant/ Purification
One of the most common and popular of ways using bleach (sodium hypochlorite) is as an disinfectant. Sodium hypochlorite is an effective and beneficial disinfectant because it denatures protein in micro-organisms and it is therefore effective in killing bacteria, fungus and viruses. ("HK Polytechnic University", 2007) Chlorine bleach is a household cleaning product often used as a disinfectant to kill germs on surface areas, such as bathtubs. ("EPA", 2014) Sodium Hypochlorite is mainly used as an disinfectant for swimming pools, it is applied in swimming pools for water purification and oxidation. ("Lenntech", 1998)
Some examples of how sodium hypochlorite are used includes:
- Water purification in personal home pools, residential pools or spas.
- Disinfectant in private wells to prevent the growth of micro-organisms.
- As a component in household cleaning agents, such as mold remover, hard surface cleaners and drain cleaners.
- Consumed in industrial water treatment systems.
(Schlager, Weisblatt, & Newton, 2006)
2. Bleaching
The second most common ways of using sodium hypochlorite is bleaching; bleaching is the process of whitening fabrics (which are commonly used for laundry purposes) and other materials. Bleaching is an example of a chemical change because a change of colour can be seen. Bleach works by the process of oxidation and due to its corrosive nature, bleach has the ability to break down the molecules of the chemical compound of a stain into smaller elements which will separate it from the fabric. ("Advameg", 2014) Bleach can be used on other materials besides fabrics, such as hair or wood.
Hair: "Once the bleach is applied to the hair, the bleach oxidizes with the melanin molecules found in hair causing it to become lighter." (Tai, 2012, para.2)
Wood: "Bleaching lightens the color of wood and reduces both the contrast of the grain lines and the contrast between sapwood and heartwood. It can be used to remove stains or dye from previous finishes." ("PaintPro", 2007, para.2)
Be cautious when using bleach because sodium hypochlorite is a highly reactive compound, it will react strongly with metals and organic materials.(Schlager, Weisblatt, & Newton, 2006) Must of the household bleach usually contains 5% sodium hypochlorite with a pH of 11. At pH 11 it only feels irritating if you come into contact with it, but at pH 13 the bleach will be corrosive and therefore will burn your skin. (Lenntech, 1998) You can also harm your respiratory system by inhaling the deadly fumes.
Watch the video below to learn how to use bleach (for laundry purposes) !