What you see above, is a video of how sodium hypochlorite can be made at home. ("UC235", 2010)
Structure
Parts
We know that the compound of sodium hypochlorite is made up of 3 elements which are; sodium, oxygen and chlorine. There is only one of each, and they each take up an even amount of space.
This is how much the compound is taken up by numbers:
Sodium: 33.3%
Oxygen: 33.3%
Chlorine: 33.3%
This is how much the compound is taken up by mass:
Sodium: 30.9%
Oxygen: 21.5%
Chlorine: 47.7%
(Makansi, 2008)
Bonding
Both ionic and covalent bonds take place to form this compound.
First, the hypochlorite ion needs to be made which is formed in a covalent bond between an oxygen and chlorine atom (shown in Figure 1). (Makansi, 2008)
Structure
Parts
We know that the compound of sodium hypochlorite is made up of 3 elements which are; sodium, oxygen and chlorine. There is only one of each, and they each take up an even amount of space.
This is how much the compound is taken up by numbers:
Sodium: 33.3%
Oxygen: 33.3%
Chlorine: 33.3%
This is how much the compound is taken up by mass:
Sodium: 30.9%
Oxygen: 21.5%
Chlorine: 47.7%
(Makansi, 2008)
Bonding
Both ionic and covalent bonds take place to form this compound.
First, the hypochlorite ion needs to be made which is formed in a covalent bond between an oxygen and chlorine atom (shown in Figure 1). (Makansi, 2008)
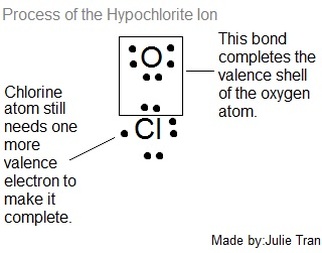
Figure 1
As you can see in Figure 1, the Oxygen atom is now neutral and complete although the Chlorine atom still needs one more valence electron to complete it's outer shell.
That's where the Sodium atom comes in which will be displayed in Figure 2.
As you can see in Figure 1, the Oxygen atom is now neutral and complete although the Chlorine atom still needs one more valence electron to complete it's outer shell.
That's where the Sodium atom comes in which will be displayed in Figure 2.
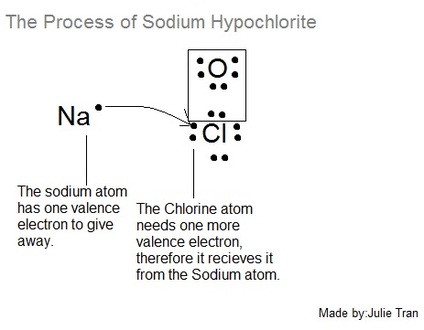
Figure 2
As you can see in Figure 2, the Sodium atom has one valence electron to give away and the Chlorine atom needs to receive one, therefore it makes a perfect ionic bond.
This combines the sodium atom to the hypochlorite ion, and this bond finalizes the bonding of the Sodium Hypochlorite atom.
As you can see in Figure 2, the Sodium atom has one valence electron to give away and the Chlorine atom needs to receive one, therefore it makes a perfect ionic bond.
This combines the sodium atom to the hypochlorite ion, and this bond finalizes the bonding of the Sodium Hypochlorite atom.
Making Sodium Hypochlorite in a Factory
In a factory like Intratec, they use chlorine and caustic soda to chemically make sodium hypochlorite. ("Intratec Solutions LLC", 2005)
Caustic soda is also known as "Sodium Hydroxide" and it's a compound that consists of sodium, oxygen and hydrogen. (Euro Chlor, 2013)Therefore, that's everything you need to create Sodium Hypochlorite with an extra hydrogen atom.
There are many stages to be done to create sodium hypochlorite. The process is split in two components called, "Reaction and Product Discharge" and "Bleach Filtration". ("Intratec Solutions LLC", 2005)
In the Reaction and Product Discharge stage, there is a chlorine absorption system that can be divided into two systems. The first system has a packed column that contains caustic safely. Then in the second system, it gets liquor from the first column and adds it to the chlorine until you get the bleach concentration that you want. ("Intratec Solutions LLC", 2005)
Water is then used to weaken the caustic soda and is mixed with the first column bottom stream which is in the first buffer tank. Then the solution goes through the first recycle pump. It cools down while doing this before it is able to reach the top of the column. The chlorine is then weakened by air and placed into the bottom of both columns. ("Intratec Solutions LLC", 2005)
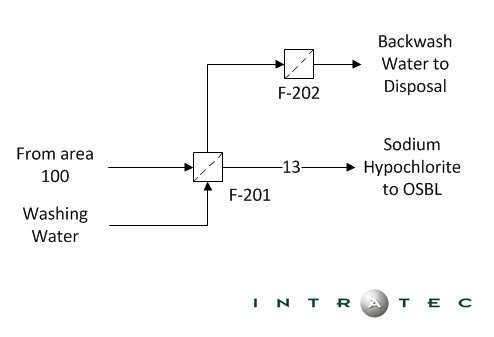
The Bleach Filtration stage is a lot like the first stage but it has it's difference as well. This stage is mandatory because it makes the product sanitary and it needs to meet quality requirements in order to be packaged and sold.
The picture below is a diagram of the Bleach Filtration stage. ("Intratec Solutions LLC", 2005)
In a factory like Intratec, they use chlorine and caustic soda to chemically make sodium hypochlorite. ("Intratec Solutions LLC", 2005)
Caustic soda is also known as "Sodium Hydroxide" and it's a compound that consists of sodium, oxygen and hydrogen. (Euro Chlor, 2013)Therefore, that's everything you need to create Sodium Hypochlorite with an extra hydrogen atom.
There are many stages to be done to create sodium hypochlorite. The process is split in two components called, "Reaction and Product Discharge" and "Bleach Filtration". ("Intratec Solutions LLC", 2005)
In the Reaction and Product Discharge stage, there is a chlorine absorption system that can be divided into two systems. The first system has a packed column that contains caustic safely. Then in the second system, it gets liquor from the first column and adds it to the chlorine until you get the bleach concentration that you want. ("Intratec Solutions LLC", 2005)
Water is then used to weaken the caustic soda and is mixed with the first column bottom stream which is in the first buffer tank. Then the solution goes through the first recycle pump. It cools down while doing this before it is able to reach the top of the column. The chlorine is then weakened by air and placed into the bottom of both columns. ("Intratec Solutions LLC", 2005)
The Bleach Filtration stage is a lot like the first stage but it has it's difference as well. This stage is mandatory because it makes the product sanitary and it needs to meet quality requirements in order to be packaged and sold.
The picture below is a diagram of the Bleach Filtration stage. ("Intratec Solutions LLC", 2005)
 The packaging of Bleach.
The packaging of Bleach.
You filter bleach for the same reason you filter water. There are things in there that you don't want to be. Unfiltered bleach contains heavy metals and unneeded solids. ("Powell", 2014)
After the filtration stage is complete, it is put into storage. Then it's packaged and sent off to markets/stores to be sold. ("Intratec Solutions LLC", 2005) It's usually packaged in a big bottle.
After the filtration stage is complete, it is put into storage. Then it's packaged and sent off to markets/stores to be sold. ("Intratec Solutions LLC", 2005) It's usually packaged in a big bottle.